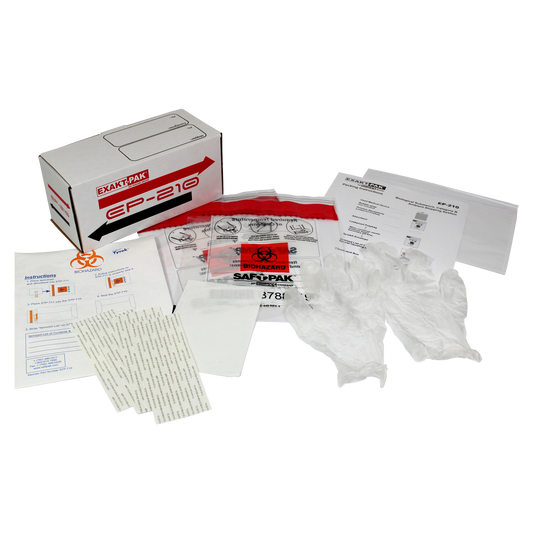
The Issue: A Potential for Exposure
The primary regulatory concern with Used Medical Devices (UMDs) is the potential for exposure to an infectious substance (a disease-causing agent). Consequently, failure to comply with the regulations as set forth by ICAO (International Civil Aviation Authority) and IATA (International Air Transport Association) can result in civil or criminal penalties.